Process Development
-
-
Upstream Process
-
We take pride in our USP team's track record of successfully cultivating a diverse range of molecules in our state-of-the art high-throughput lab. Our team of experts is committed to delivering high-quality results at speed and has extensive experience in optimizing upstream process development to meet the unique needs of our clients.
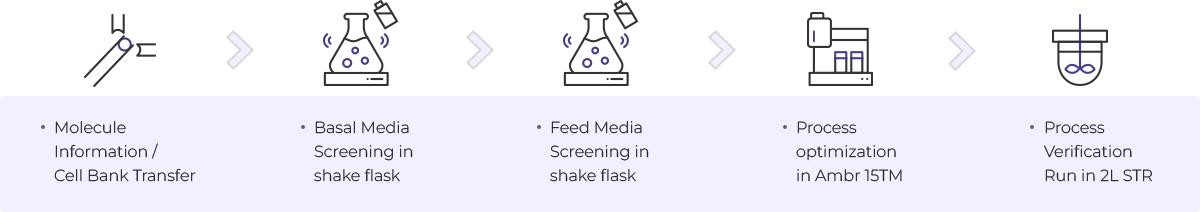
Upstream Process
- Molecule information / cell bank transfer
- Basal Media Screening in shake flask
- Feed Media Screening in shake flask
- Process optimization in Ambr 15TM
- Process verification run in 2L STR
-
 Discover your molecule’s hidden potential
Discover your molecule’s hidden potential
 offers the following services:
offers the following services:
- Media and/or additive screening by optimizing cell culture conditions
- Process parameter optimization by selecting best quality attribute conditions
- Investigation of ADCC/CDC* through substantial comparability studies & CQAs determined based on analyses of extensive data
- Pre-integrated services
-
-
Efficient execution under
a systemized regimen
-
Efficient execution under
- Optimal decision-making
-
-
DoE insights
with ADCC & CDC
-
DoE insights
- Client-centric adjustments
-
-
N-glycan modulation
at client’s will
-
N-glycan modulation
Connect with our development experts
-
-
Downstream Process
-
Our DSP team is equipped with pioneering lab capabilities and up-to-date techniques. In addition to our expertise in DSP, we have also developed a streamlined scheme for non-GMP toxicology manufacturing, help steering your development journey.
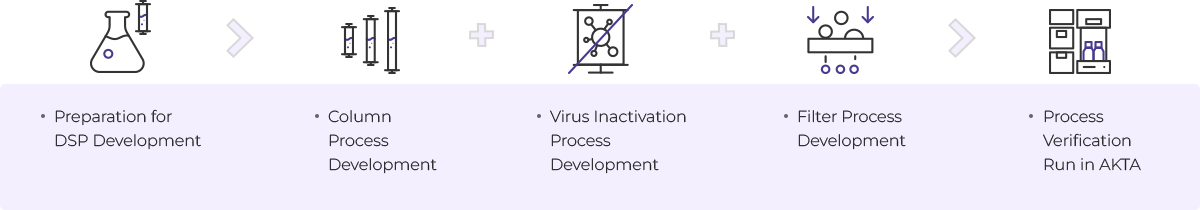
Downstream Process
- Preparation for DSP Development
- Column Process Development
- Virus Inactivation Process Development
- Filter Process Development
- Process verification run in AKTA
-
-
-
Formulation
-
Our formulation team is filled with high-quality subject matter experts (SMEs) who are leading the latest techniques and industry trends. Our commitment to exceeding our customers’ expectations is at the forefront of everything we do.
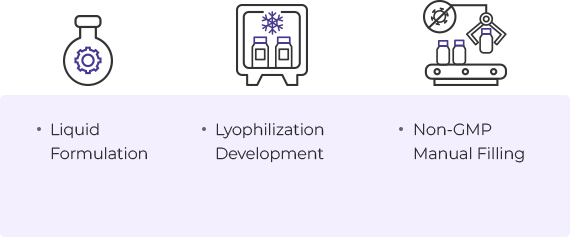
Formulation
- Liquid Formulation
- Lyophilization Development
- Non-GMP Manual Filling
-
Analytical Method
-
With a well-developed strategy for method development and a high performance, our analytical method development team is dedicated to delivering reliable results possible. We use state-of-the-art instrumentation and the latest techniques to ensure the quality of our analytical data.

Analytical Method
- A nalytical Method Development
- Extended characterization & comparability studies
- Analytical Support
-
Process Development
- Upstream Process
-
We take pride in our USP team's track record of successfully cultivating a diverse range of molecules in our state-of-the art high-throughput lab. Our team of experts is committed to delivering high-quality results at speed and has extensive experience in optimizing upstream process development to meet the unique needs of our clients.
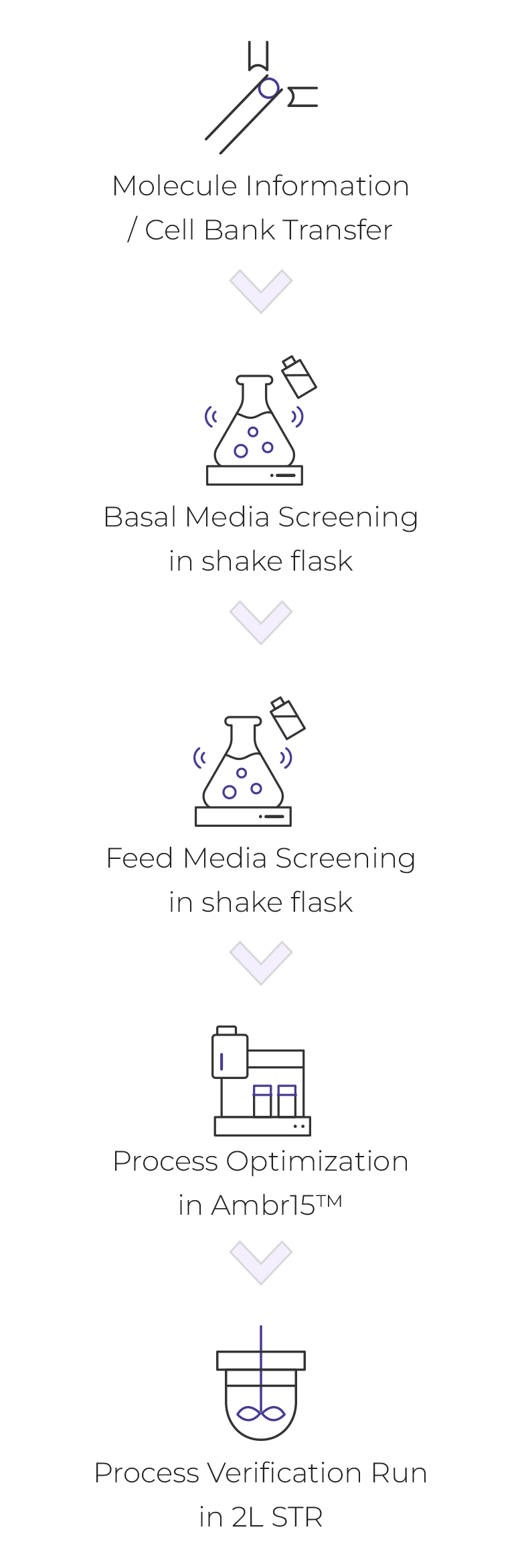
Upstream Process
- Molecule information / cell bank transfer
- Basal Media Screening in shake flask
- Feed Media Screening in shake flask
- Process optimization in Ambr 15TM
- Process verification run in 2L STR
 Discover your molecule’s hidden potential
Deep understanding of a molecule's unique charactertistics is crucial. Samsung Biologics' development platform S-Glyn™ provides modulation of N-glycan, one of the potential critical quality attributes, via in-depth investigation at your will. Unlock the true potential of a molecule and steer your development with S-Glyn™.
Discover your molecule’s hidden potential
Deep understanding of a molecule's unique charactertistics is crucial. Samsung Biologics' development platform S-Glyn™ provides modulation of N-glycan, one of the potential critical quality attributes, via in-depth investigation at your will. Unlock the true potential of a molecule and steer your development with S-Glyn™. offers the following services:
offers the following services:
-
Media and/or additive screening
by optimizing cell culture conditions -
Process parameter optimization
by selecting best quality attribute conditions -
Investigation of ADCC/CDC*
through substantial comparability studies & CQAs determined based on analyses of extensive data
* ADCC: Antibody-dependent cellular cytoxicity / CDC: Complement-dependent cytoxicityConnect with our development experts
- Downstream Process
-
Our DSP team is equipped with pioneering lab capabilities and up-to-date techniques. In addition to our expertise in DSP, we have also developed a streamlined scheme for non-GMP toxicology manufacturing, help steering your development journey.
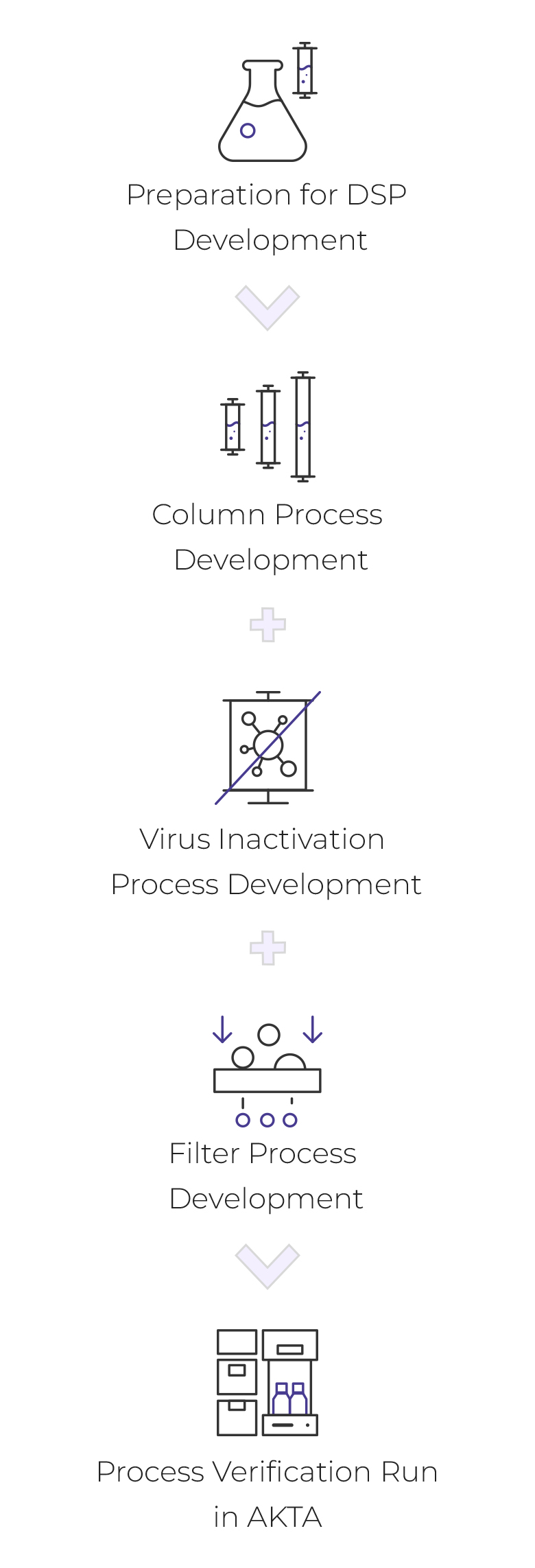
Downstream Process
- Preparation for DSP Development
- Column Process Development
- Virus Inactivation Process Development
- Filter Process Development
- Process verification run in AKTA
- Formulation
-
Our formulation team comprises plenty of high-quality subject matter experts (SMEs) who are well-versed in the latest tech-niques and industry trends. Our commitment to exceeding our customers’ expectations is at the forefront of everything we do.

Formulation
- Liquid Formulation
- Lyophilization Development
- Non-GMP Manual Filling
- Analytical Method
-
With a well-developed strategy for method development and a high performance, our analytical method development team is dedicated to delivering reliable results possible. We use state-of-the-art instrumentation and the latest techniques to ensure the quality of our analytical data.
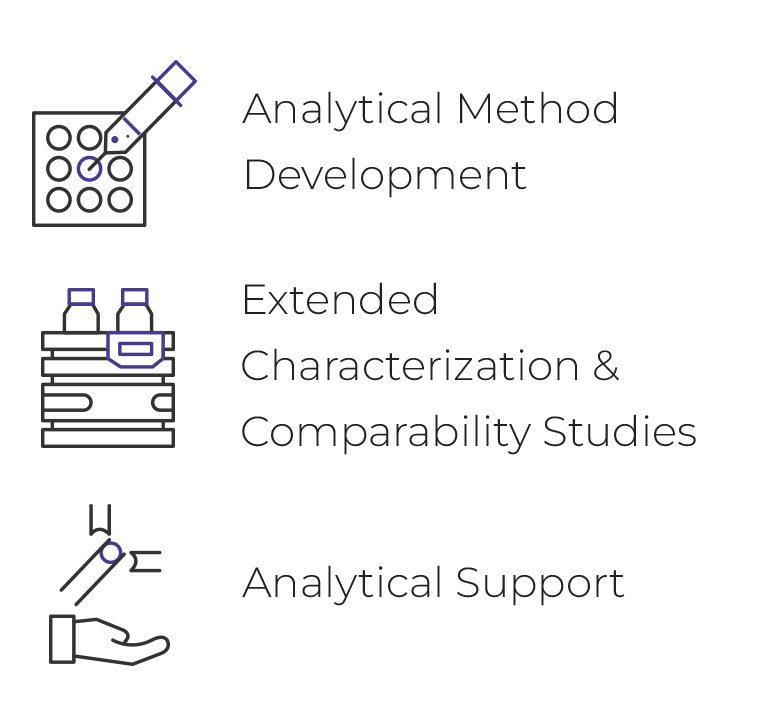
Analytical Method
- A nalytical Method Development
- Extended characterization & comparability studies
- Analytical Support

